
The nuclei of most atoms also contain neutrons. Differentiate between atomic number and mass number. Where is nearly all of the mass of an atom located Explain your answer in terms of what contributes to this mass. 
"We're witnessing a period of discovery similar to the 1950s, when a 'particle zoo' of hadrons started being discovered and ultimately led to the quark model of conventional hadrons in the 1960s. The nuclei of all atoms contain subatomic particles called protons. What are the names, symbols, electrical charges, and relative mass of the three subatomic particles 2. "The more analyses we perform, the more kinds of exotic hadrons we find," physicist Niels Tuning said in a statement. According to modern atomic theory, an atom has a nucleus, which is its center, or core. More rarely, however, they can also combine into four-quark and five-quark particles, or tetraquarks and pentaquarks. A subatomic particle is a unit of matter or energy thats the fundamental makeup of all matter. Quarks are elementary particles that usually combine in groups of twos and threes to form hadrons such as the protons and neutrons that make up atomic nuclei. 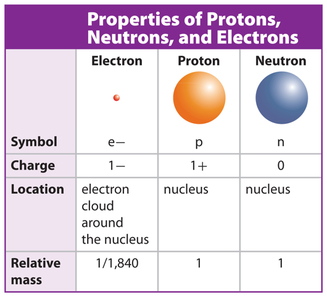
They will help physicists better understand how quarks bind together into composite particles. As a result, the existence of various types of matter around us is due o the presence of atoms in them. Atoms are the fundamental components of matter. Protons, electrons, and neutrons are the three subatomic particles that typically make up an atom. These subatomic particles include protons, electrons and. A subatomic particle is a particle a size smaller than that of an atom. Dozens of subatomic particles have been discovered. In this video, you will learn the properties of the three subatomic particles relevant in chemistry. An electron is an example of an elementary particle protons and neutrons are examples of composite particles. Now scientists at CERN say they have observed a new kind of "pentaquark" and the first-ever pair of "tetraquarks", adding three members to the list of new hadrons found at the LHC. A subatomic particle is a particle smaller than an atom.It may be either an elementary (or fundamental) particle, or a composite particle, also called a hadron. The 27 kilometre-long (16.8 mile) LHC at CERN is the machine that found the Higgs boson particle, which along with its linked energy field is thought to be vital to the formation of the universe after the Big Bang 13.7 billion years ago. A neutron's mass is slightly larger than that of a proton.GENEVA, July 5 (Reuters) - Scientists working with the Large Hadron Collider (LHC) have discovered three subatomic particles never seen before as they work to unlock the building blocks of the universe, the European nuclear research centre CERN said on Tuesday.They are uncharged particles found within all atomic nuclei (except for hydrogen).Elements are arranged in the Periodic Table of the Elements in order of increasing atomic number. The three subatomic particles determine the overall charge of an atom, the chemical characteristics it can possess and its physical properties.The number of protons in an atom is referred to as the atomic number of that element.The charge of the proton is +1.602 x 10 -19 Coulomb.They are inside the nucleus of the atom. Here you will find the General Science Multiple Choice Questions Mcqs are from Biology, Chemistry, Physics and Atmospheric Studies.The number of filled electron shells increases as moving down a group in the periodic table.The charge of the electron is -1.602 x 10 -19 Coulomb.It has a radius 2.5 times larger than a proton. The radius of the electron is 2.82 x 10-15 m.According to the Standard Model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a proton, neutron, or meson), or an elementary particle, which is not composed of other particles (for example, an electron, photon, or muon). The number of electrons in an atom largely decides the properties of the atom. In physics, a subatomic particle is a particle smaller than an atom. 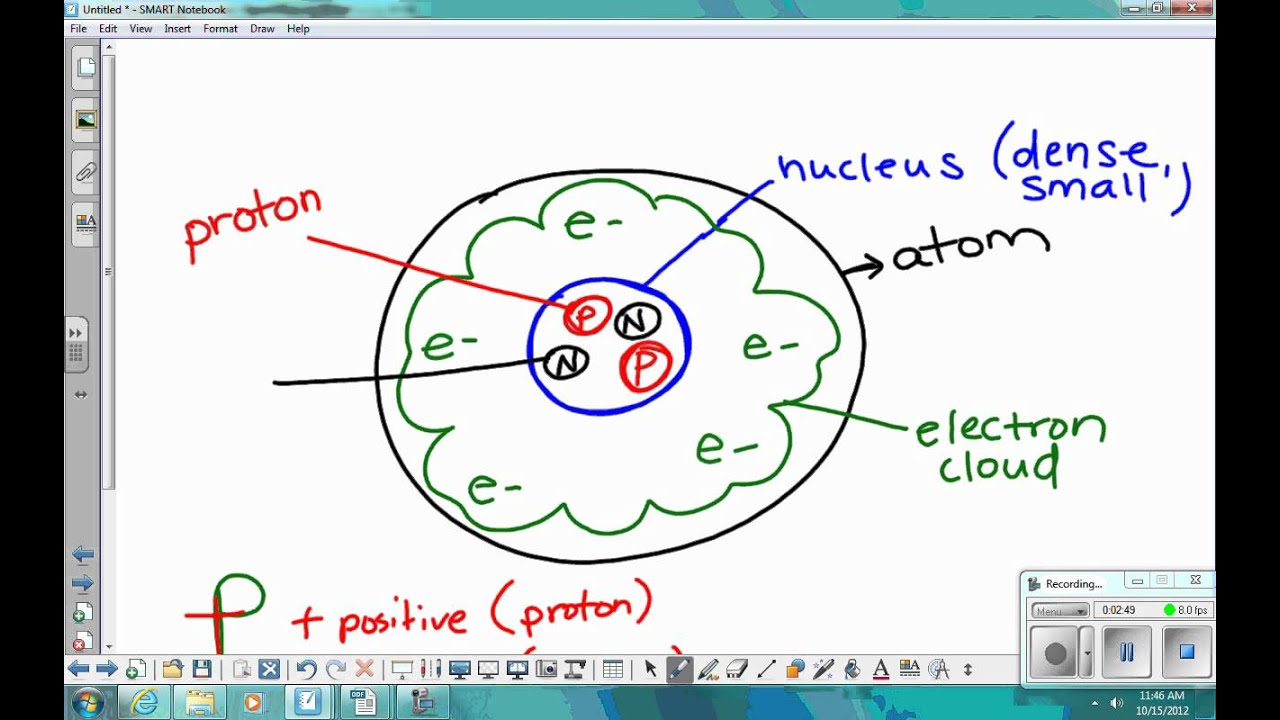
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
